Sphere-shaped landmark relies on well-rounded coating system
Think zinc
Zinc-rich primers have a history of success in providing corrosion protection to bridges, potable water storage tanks, and architecturally exposed structural steel—even those subjected to harsh environmental and corrosive elements. This is because when a zinc-rich primer cures and hardens, it creates a reactive barrier, protecting the steel substrate from weathering. As the degradation rate of zinc is many times less than that of steel, the zinc will sacrifice itself, thus protecting the underlying steel. (Find this information in K. Nanan’s 2017 Corrosionpedia article, “How Metallic Coatings Protect Metals from Corrosion.”)
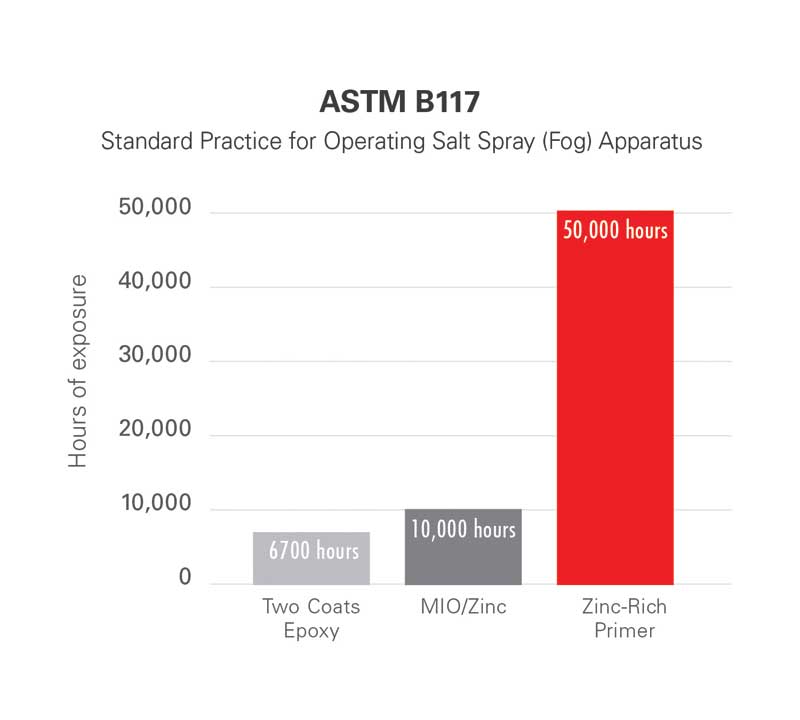
The primer used in the spheres project contains 83 percent zinc pigment by weight in the cured film and provides sacrificial (or galvanic) protection should the structural steel become scratched or damaged during construction. The performance of this primer has been demonstrated by the coating manufacturer—both in the field and within the laboratory—using accelerated testing in accordance with ASTM B117, Standard Practice for Operating Salt Spray (Fog) Apparatus.
Salt spray testing
The coatings manufacturer used a standard salt spray (or fog) test, where sample panels were coated with the zinc-rich primer and placed in a controlled chamber. Intervals of hot, de-ionized water were released into the enclosed space to create humid conditions. Inside the salt spray (fog) apparatus, the specimens were exposed to a neutral (pH 6.5 to 7.2) salt fog solution, containing five percent sodium chloride, at 35 C (95 F). This acted as an accelerant to induce corrosion. (To read more, refer to “Testing Remains Relevant for Coating Selection” by M. Thomas in the July 2011 edition of The Construction Specifier. The definition is derived from Corrosionpedia, “What does ASTM B117 mean?”)
Next, to simulate what occurs when a topcoat is scratched or damaged at a construction site, testers cut a scribe through the coating film and exposed the metal substrates to the chloride solution. This component of the test had to be conducted because undercutting can result in the coating lifting and peeling away from the steel substrate, opening the surface to severe corrosion.
After 50,000 hours of exposure to the salt fog conditions in the controlled chamber, specimens coated with the primer showed no blistering, cracking, or delamination, little undercutting (or creepage) at the scribe, and no more than one percent rusting. For comparison, metal samples protected solely by two coats of epoxy were subjected to the same test and lasted only 6700 hours in the controlled chamber, while a micaceous iron oxide (MIO)/zinc prime coat exposed to 10,000 hours revealed 0.4-mm (0.016-inch) creepage and three percent rusting at the scribe (Figure 1).
Fluoropolymer finish
The proposed fluoropolymer finish coating also underwent accelerated weathering testing to verify its durability and aesthetic performance. Under testing conducted by the coating manufacturer in accordance with ASTM D4587, Standard Practice for Fluorescent UV-Condensation Exposures of Paint and Related Coatings, the chemistry found in this fluoropolymer technology showed almost no color or gloss change after 10,000 hours of accelerated UV exposure. (ASTM D4587, Standard Practice for Fluorescent UV-Condensation Exposures of Paint and Related Coatings, is available at www.astm.org/DATABASE.CART/HISTORICAL/D4587-05.htm.)
The finish coat was also subjected to a test known as the equatorial mount with mirrors for acceleration with water (EMMAQUA)—a particularly harsh outdoor accelerated weathering test in accordance with ASTM D4141, Standard Practice for Conducting Black Box and Solar Concentrating Exposures of Coatings, designed to simulate wet climates. Metallic specimens coated with the fluoropolymer finish were exposed to UV energy, which was measured in megajoules per square meter (MJ/m2), to determine levels of gloss, color, and any physical degradation. When exposed to 3500 MJ/m2, the tested fluoropolymer coating showed no blistering, cracking or chalking, slightly less color change, and significantly better gloss retention than a standard acrylic polyurethane.
Both accelerated weather tests were consistent with the aesthetic performance of fluoropolymer coating systems observed from weathering tests conducted in South Florida. (This is based on proprietary testing conducted by the coatings manufacturer.)The aesthetic reliability of these coatings has also been supported by the research of trained coating specialists who have observed ambient-cured fluoropolymer coatings on both new and maintenance landmark architectural projects over the past 15-plus years under a wide range of exposure conditions, which include high-humidity coastal environments, desert climates, and areas with frequent freeze-thaw cycles. (This is based on anecdotal observations of manufacturer coating consultants and research and development specialists.)




